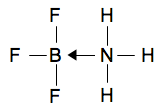|
Chemguide: Core Chemistry 14 - 16 Dative covalent (co-ordinate) bonding This page looks at the formation of covalent bonds where both of the bonding electrons come from the same atom. Your syllabus may not mention this, but it is important in understanding work on acids and bases later. It is also not in the least bit difficult! I am assuming that you have read the page introducing covalent bonding and have access to a Periodic Table such as the one you can download from this site. The download button is at the beginning of the second paragraph under the table. | |
|
Note: You will probably come across an alternative spelling of co-ordinate. It is frequently written without the hyphen: coordinate. Both are commonly accepted and I know that I am inconsistent about which I use. I am going to stick to the easier name of dative covalent bonding for now. | |
|
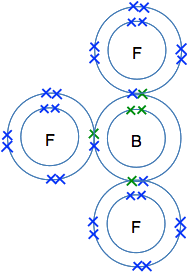
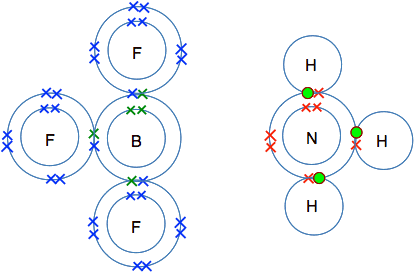
The reaction between BF3 and NH3 This is not a reaction that you need to remember or even know about at this level, but it is the simplest and most easily understood example of dative covalent bonding. So I am going to talk about it anyway. BF3 is boron trifluoride and NH3 is ammonia. You will already have met the bonding in ammonia on a previous page. So we will start by looking at the bonding in BF3. Boron's atomic number is 5, and so it has an electronic structure of 2,3. There are three single electrons which can be paired up to form bonds by sharing with an unpaired electron in each of three fluorine atoms.
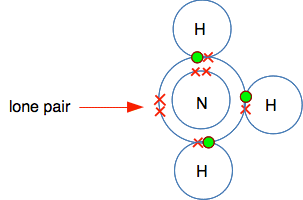
BF3 is known as an electron deficient compound, because boron's outer level has room for two more electrons - but boron doesn't have any more electrons for anything else to share with. So let's look at ammonia. The next diagram isn't quite the same as the one you have seen before, because I have rotated it through 90° for reasons that will become clear in a moment.
The really important bit of this structure is the lone pair in the outer level. Remember that a lone pair is a pair of electrons not involved in a bond. For reasons that you will discover if you do chemistry at a higher level, the lone pair in ammonia is quite active - it tends to involve itself in bonding. If you line up these two molecules next to each other, it is obvious what is going to happen.
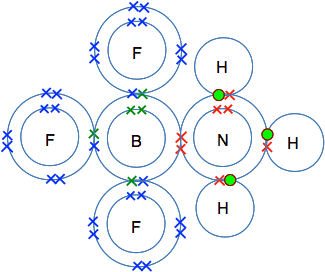
There is an empty space in the outer level of the BF3, and a lone pair on the nitrogen in the ammonia. If they got close together so that the lone pair filled the space, then you have created a new bond.
Creating bonds releases energy, and so the new compound formed is going to be more energetically stable than the two smaller molecules on their own. The bond formed - where both of the bonding electrons come from the same atom - is known as a dative covalent or co-ordinate covalent bond. Dative covalent bonds are sometimes (although not always) shown by drawing the bond as an arrow rather than as a simple line. The arrow starts from the atom which is donating the lone pair and points towards the atom receiving it.
Important: I repeat that this example is just to show you how a dative covalent bond is formed using an empty space in the outer level of one atom, and a lone pair on another one. As long as you understand that, you can probably safely forget the details of this reaction. You will need to come back to this when we look at acid-base reactions. I will remind you to read it again at the right time.
© Jim Clark 2019 |
|