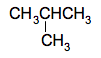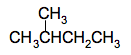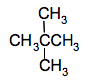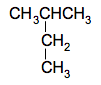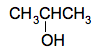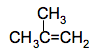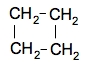|
Chemguide: Core Chemistry 14 - 16 A short introduction to structural isomerism The title perhaps sounds scary but you will already have met this if you have read the pages about organic formulae and organic names, although we haven't used the term yet. If you haven't read those, you will find this a lot easier if you read them first. What are structural isomers? Remember that the molecular formula just counts the number of each sort of atom in the compound. A structural formula shows how these are combined together. In structural isomerism, you can find more than one way of joining up the atoms to give different molecules. These different structures which can be made from the same combination of atoms are called structural isomers. Structural isomerism in the alkanes The alkanes are a group of hydrocarbons which we will look at in more detail later. For now we are only interested in the structure of their molecules. The first 6 alkanes are:
Now imagine you were making models of these. In methane, ethane and propane there is only one possible way of joining up the necessary numbers of atoms so that each carbon is forming 4 bonds and each hydrogen is only forming one bond. CH4 CH3CH3 CH3CH2CH3 Isomerism in butane But butane, C4H10, is different. It is actually possible to make two different molecules from this same combination of atoms. They are CH3CH2CH2CH3 which is just called butane; and
which is called 2-methylpropane. These are structural isomers - they have the same molecular formula but different structural formulae. | |||||||||||||
|
Important: If you haven't got ways of drawing organic molecules or the names of organic molecules sorted out, go back and get it sorted now. The longer you put it off, the more difficult you are making it for yourself. | |||||||||||||
|
You can tell if you have got structural isomers by imagining you have a model of one of them. Can you turn it into the other one just by twisting it or rotating the molecule about the single bonds? If you can't - if you have to pull the model to pieces and re-make it - then you have structural isomers.
There is no way you can just rotate or twist a model of butane to turn it into 2-methylpropane. Isomerism in pentane It is fairly unlikely that you will have to work out isomers of pentane at this level, but it isn't difficult. Pentane has a molecular formula C5H12. Start by drawing the longest chain horizontally: CH3CH2CH2CH2CH3 Now work systematically. Take a CH3 group (a methyl group) off the end of the chain to leave a 4-carbon chain, and see where else you could fit it in place of one of the other hydrogens. The hydrogen, of course, would be used to join to the carbon you took the methyl group off. You would come up with this:
It doesn't matter whether you draw the methyl group pointing upwards or downwards. It would be the same molecule - all you are doing is rotating around a carbon-carbon bond. This is a 4-carbon chain with a methyl group attached to the number 2 carbon. The name is 2-methylbutane. There is nowhere else you could attach the methyl group which would produce a different molecule. If, for example, you attached it to the other middle carbon instead, that would be exactly the same structure, just flipped over. Now take another methyl group off the end of the chain to leave a 3-carbon chain so that you now have 2 methyl groups to position. There is only one way of doing that - by attaching both of them separately to the middle carbon atom.
That's called 2,2-dimethylpropane. You name the position of both of the methyl groups. And those 3 structures above are the only structural isomers of pentane. Beware! You might think that you could take the two methyl groups that you have taken of the end of the chain and make them into one CH3CH2 which you could attach to the middle carbon of the 3-carbon chain.
We have already talked about this on the previous page. Your longest chain in this is no longer horizontal. You have a bent 4-carbon chain. Straighten it out and you have a 4-carbon chain with a methyl group attached to a next-to-the-end carbon. We have already got that structure drawn above. This is why when you are working out structural isomers like this you always have to check that your longest chain is still the horizontal one. | |||||||||||||
|
Note: At this level you are quite likely to need to know about the two structural isomers of butane. Check your syllabus / specification to find out exactly what else you need to be able to do. | |||||||||||||
|
A couple of other simple examples of structural isomerism This is essentially just a repeat of stuff you will already have read on the names of organic molecules page, except that at that point I didn't call them structural isomers. Structural isomerism in alkenes Alkenes are like alkanes, except that they contain a carbon-carbon double bond. The "en" code in the name tells you that there is a carbon-carbon double bond. The simplest two are ethene, CH2=CH2, and propene, CH3CH=CH2 - neither of which has any isomers which still contain a double bond. But butene, with 4 carbon atoms, does have structural isomers. The ones you might possibly come across at this stage are
The position of the double bond in the chain has changed. The numbers show the position of the carbon that the double bond starts on. Remember that you always choose the lowest possible numbers in a name. The "1" shows it starts on an end carbon; the "2" shows it starts on a next-to-the-end carbon. | |||||||||||||
|
Note: There are two other structural isomers of C4H8 as well. One of them also contains a carbon-carbon double bond; the other doesn't. It isn't important that you should find them, but if you want to try, you will find their structures at the bottom of the page. | |||||||||||||
|
Structural isomerism in alcohols Alcohols are compounds which contain an OH group attached to a carbon chain. The commonest one is ethanol, CH3CH2OH, and you will meet that in more detail later. If you have read the page about the various ways of writing organic formulae, you will have come across an isomer of ethanol with the structure CH3OCH3. That isn't an alcohol because it doesn't contain an OH group. We didn't call it a structural isomer at the time, but that's what it is - a compound with the same molecular formula, C2H6O, but a different structural formula. What I want to look at now are isomers of alcohols which are themselves alcohols. This starts to happen with propanol, C3H8O, and I just want a quick look at that. There are two different alcohols with that formula. The obvious one is CH3CH2CH2OH This is called propan-1-ol. The "1" shows that the OH group is on the number 1 carbon atom - a carbon atom at the end of the chain. But you could equally well put the OH group in the middle of the chain to give propan-2-ol.
If you have read the page about naming organic compounds, you will have read the same sort of discussion of butan-1-ol and butan-2-ol. These again are structural isomers. | |||||||||||||
|
Note: There are actually two other alcohols which are structural isomers of butanol as well. I'm not going to clutter this page any further, but if you wanted to try to find them, you can easily check your answer by googling structural isomers of butanol. | |||||||||||||
|
The other two structural isomers of C4H8 (see above) There is one with a carbon-carbon double bond, and also a branched chain,
and one where the carbon atoms are joined up in a ring.
Very well done indeed if you got the second one.
© Jim Clark 2021 |
|||||||||||||